
© Blauplanet.com
Other sections:




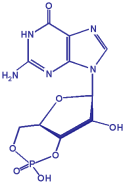
Cyclic GMP
Advanced
Created by Dr. Luis Agulló (last update on 22-4-2008)
Soluble Guanylyl/Guanylate Cyclase: Structure and Nitric Oxide Activation
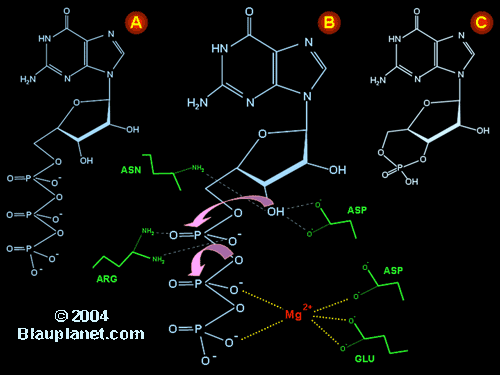
Fig. 1. Catalysis of cyclic GMP synthesis by soluble guanylyl cyclase.
Nitric oxide sensitive guanylyl cyclases or 'soluble' guanylyl cyclases are heterodimers composed of two different subunits: α (73-82 kDa) and β (70-76 kDa). α and β have limited homology in their sequence, but similar domains can be distinguished: an amino terminus domain, a center domain, and a catalytic domain (Figure in preparation). The sequence of aminoacids that participate in the catalytic reaction are located in the C-terminus, while the N-terminus has a regulatory role and is responsible for the sensitivity to nitric oxide. The central domain seem to be implicated in the interaction between α and β subunits to form the heterodimer. Several isoforms of these subunits have been described: α1, α2 and αi2 (splice variant of α2) and β1, β2 and β3. The heterodimer α1β1 is practically present in all tissues studied. Other heterodimer described is the combination α2β1. This one has been only found in important quantities in the brain (where the concentration of α1 and α2 is similar) and in some fetal tissues. The existence of other possible combinations is less clear. Although inactive heterodimers have been obtained in cells transfected with the β2 subunit and an α subunit, demonstration of the presence of this heterodimer in vivo remains elusive.
As commented in the introduction to guanylyl cyclases, new data suggest that both active heterodimers presently known, α1β1 and α2β1, could associate to the cell membrane, where they might localize close to nitric oxide synthases. This will be discussed in more detail in another section of this site (in preparation).
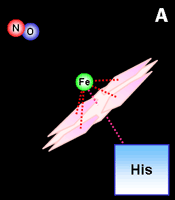 |
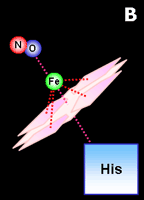 |
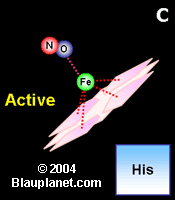 |
Fig. 2. Activation by nitric oxide.


Author: Dr. Luis Agulló (luis.agullo [at] lagullo.com).
Edited by Blauplanet.com, Cerdanyola del Vallès, Barcelona, Spain.
Webmaster: info [at] blauplanet.com. Conditions of use.